Parents are increasingly alarmed as cases of botulism in infants linked to ByHeart baby formula have emerged, with questions arising regarding why earlier reports were not investigated as part of the ongoing outbreak. Health officials have confirmed contamination in the formula, prompting a more extensive inquiry into the illness’s origins and potential reach.
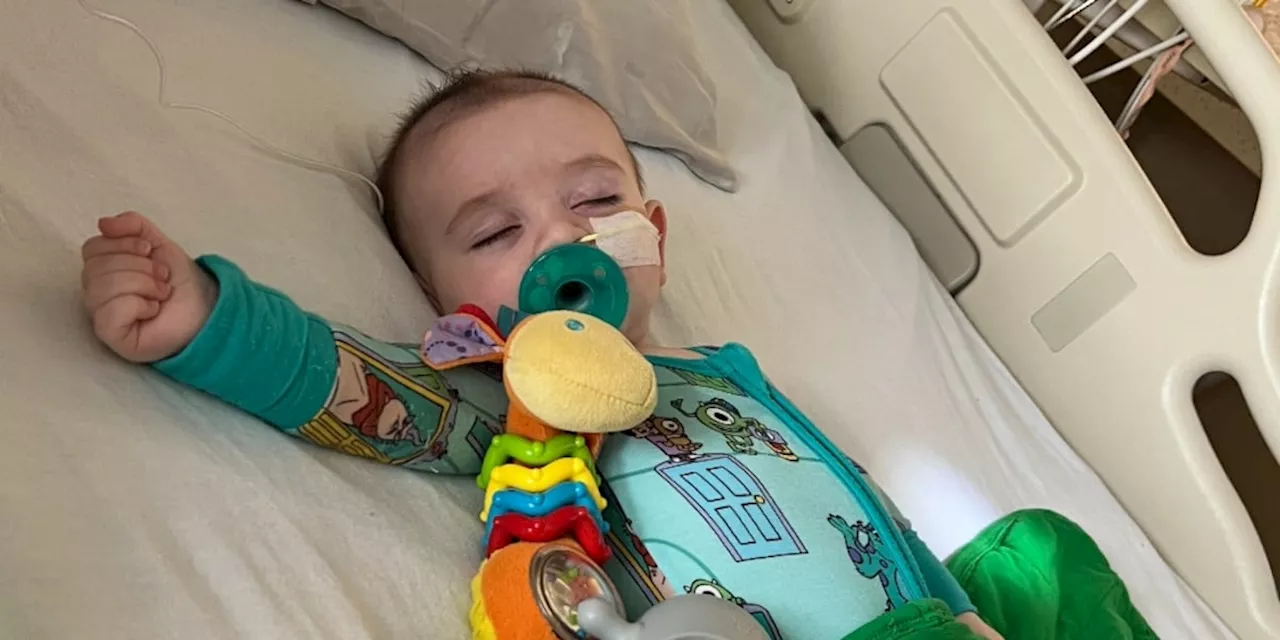
Between November 2024 and June 2025, six infants in California who consumed ByHeart formula were treated for botulism, significantly predating the outbreak that has since affected at least 31 babies across 15 states. The California Department of Public Health noted that, at the time, there was not enough evidence to identify a common source for these earlier cases.
According to reports received by The Associated Press from families represented by food safety attorney Bill Marler, at least five other infants were treated for the rare and potentially life-threatening disease after ingesting ByHeart formula in late 2024 and early 2025. Among those affected is Amy Mazziotti of Burbank, California. Her son, Hank, fell ill in March 2025, just weeks after starting ByHeart. Similarly, Katie Connolly from Lafayette, California, reported that her daughter, M.C., was hospitalized for botulism in April 2025 after being fed the formula to help with her sleep. Both mothers were initially unaware of the possible link to the product.
ByHeart officials confirmed that laboratory analyses of unopened samples of their formula revealed contamination with the bacteria that causes infant botulism. Marler has indicated that at least three additional cases, occurring before the current outbreak, involved infants who consumed the formula and received treatment for the illness.
Dr. Jennifer Cope, leading the investigation at the U.S. Centers for Disease Control and Prevention (CDC), acknowledged that there was awareness of earlier illness reports. However, she emphasized that current efforts are concentrated on understanding the surge in cases documented since August 1, 2025. Cope pointed out the complexities in definitively linking earlier cases to the outbreak, citing the time lapse and the likelihood that parents may lack records of product lot numbers or retain empty cans.
Connolly expressed her frustration, questioning why the investigation was triggered by cases originating in August while earlier cases were overlooked. Health officials explained that the clear connection between ByHeart and infant botulism cases only became evident in recent weeks. Prior to the current outbreak, no powdered infant formula in the U.S. had tested positive for the botulism-causing bacteria, and the number of cases had previously fallen within an expected range.
The identification of the botulism-causing germ in a sample from a can of ByHeart formula, in conjunction with the increase in cases—particularly on the East Coast and among very young infants—prompted notifications to the CDC, the U.S. Food and Drug Administration (FDA), and the public.
Infant botulism, while rare, poses a serious risk to health when babies ingest spores that germinate in their intestines and produce toxins. The bacterium responsible is naturally found in the environment, often making the source difficult to trace. The California Infant Botulism Treatment and Prevention Program is actively monitoring reports of the illness and the distribution of the only available treatment, an intravenous medication known as BabyBIG.
Food safety experts, including Frank Yiannas, former deputy commissioner for food policy and response at the FDA, advocate for the inclusion of earlier cases in the outbreak count if the infants consumed ByHeart formula and required treatment for botulism. He questioned the rationale behind excluding these cases.
As the investigation into the ByHeart infant formula botulism outbreak continues, authorities are focused on determining the full extent of the contamination and preventing further illnesses. The emphasis remains on thoroughly investigating the timeline of the outbreak and identifying potential sources of contamination within the ByHeart formula. Families affected by this situation are left to navigate the trauma of their children’s illnesses while seeking clarity on the safety of a product they once relied upon.