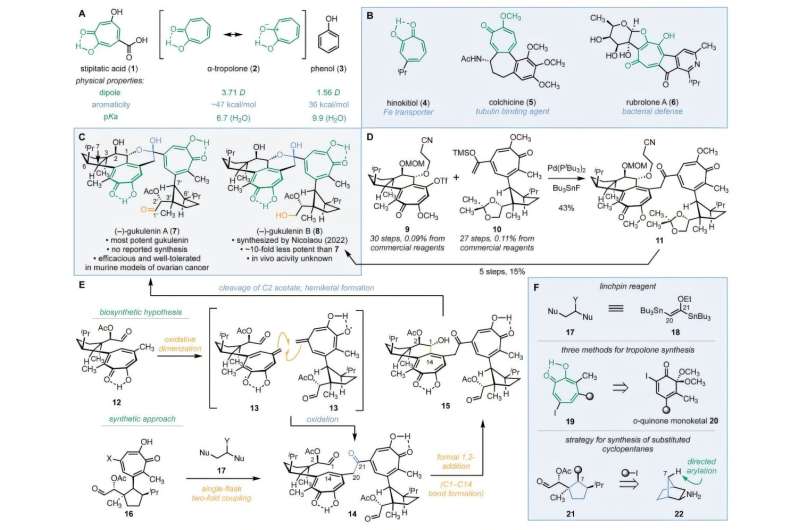
Researchers from Yale University have successfully completed the first stereoselective synthesis of the complex natural product (–)-gukulenin A, known for its significant cytotoxic effects against ovarian cancer. This breakthrough, reported on November 18, 2025, addresses long-standing challenges in the synthesis of this molecule, which has presented difficulties due to its intricate molecular architecture.
The synthesis of (–)-gukulenin A has proven particularly daunting because of its unique features, including two α-tropolones, ten 3D stereocenters, and sensitive chemical groups such as a hemiketal and an aldehyde. To overcome these challenges, the research team employed a novel three-component assembly strategy inspired by natural biosynthetic pathways.
Innovative Three-Step Strategy
The synthesis process involved three key steps: constructing the two halves of the molecule, connecting those halves, and closing the final rings. Initially, the researchers utilized exo-2-norbornylamine, an easily accessible bicyclic compound that directed the three-dimensional arrangement of the structure. They then developed a new ring-expansion technique to convert a six-membered ring into the required seven-membered tropolone structure.
The monomers were joined using a pioneering two-carbon linking reagent, specifically (E)-1,2-di(tributylstannyl)-1-ethoxyethylene. The final step involved closing the delicate hemiketal ring, achieved by simply heating the intermediate to 120 °C, resulting in a precise reaction that completed the synthesis of (–)-gukulenin A.
After successfully producing this compound, the team went on to design 15 additional derivatives. They evaluated the cytotoxicity of these derivatives across four human cancer cell lines: lung, colon, leukemia, and ovarian. Their findings indicated that derivatives containing dimeric α-tropolone rings exhibited at least ten times the potency compared to their monomeric counterparts, with some being up to 200 times more effective.
Potential for Cancer Treatment
The origins of (–)-gukulenin A can be traced back to the marine sponge Phorbas gukulensis, collected near Gageodo Island, South Korea. Since the 1950s, researchers have been intrigued by α-tropolones, leading to the extraction of various derivatives from tree barks, flowers, and bacteria. The biological activity of the gukulenins, particularly (–)-gukulenin A, has been remarkable; studies in mice have shown that it reduced ovarian tumor sizes by more than 92%. Unlike many other cytotoxic compounds, it demonstrated a selective action and was well-tolerated in animal models.
The ability to synthesize (–)-gukulenin A at scale could significantly advance therapeutic options for ovarian cancer. The researchers believe that the cytotoxic potency may stem from α-tropolone’s affinity for divalent metals, allowing (–)-gukulenin A to bind to two separate metal-containing proteins simultaneously. These insights are expected to facilitate the identification of the molecular targets of (–)-gukulenin A and pave the way for preclinical evaluations of its synthetic derivatives as potential anticancer agents.
The findings from this study are published in the journal Science. This research not only sheds light on the intricate chemistry of (–)-gukulenin A but also highlights the ongoing quest for new cancer treatments that can provide hope to patients facing challenging diagnoses.